EBAMed, a Geneva-based startup, is patenting a compact radiation field imaging device meant to boost the precision of radiation therapies. Greater precision, when it comes to dealing with the double-edged sword of radiation, translates to greater safety.
Radiotherapy is an advanced medical tool used to treat cancer and ease cancer symptoms. However, the human body can only safely absorb a certain amount of radiation over a lifetime: a federal advisory committee recommends limiting exposure to 1,000 millirems multiplied by a person’s age.
A millirem is 1/1000th of a rem, the unit measure for ionized radiation. For reference, the average American receives, just from their environment, a radiation dose of about 620 millirems each year; there is 10 millirem in a chest X-ray, but 5 million millirems for cancer treatment. It takes about a radiation dose of 450,000 millirem across the entire body for it to be lethal. Beaming radiation onto a specific tumor site thus makes radiotherapy relatively safe.
Radiation can be as harmful as it is helpful, which is why exposure to it in healthcare settings is sought to be limited wherever possible. EBAMed’s patent application is aimed at making a safer radiotherapy approach even safer through the use of hadron therapy. Unlike X-rays, it uses particles that can penetrate tissue with little diffusion, depositing the maximum energy just before stopping. The Bragg peak serves as a clear-cut guide for targeted radiotherapies, and not just those involving cancer.
The Swiss company is named after EBA or external beam ablation, a form of radiotherapy that scars heart tissue to block abnormal electrical signals leading to arrhythmias, or rapid and irregular heartbeats. A way of monitoring radiotherapy’s effects in real time can prove valuable where the heart is at risk of collateral radiation damage.
EBAMed’s creation is an imaging instrument that continuously records the position and dose of irradiation imparted to a patient during a hadron therapy session. The device is expected to enable real-time interactive corrections to treatment plans. It specifically monitors gamma rays. While medical equipment already exists for that purpose, EBAMed says its invention differs in that it is light and compact.
The Prompt Gamma Monitor EBAMed describes in its USPTO filing provides, in real time, 2-D images of the prompt gamma fields released during patient treatment. It works via two detection modules, each in the form of a LYSO or lanthanum bromide scintillating crystal. Scintillators convert radiation—in this case, gamma rays—into measurable visible light. A module may be an assembly of closely packed and optically insulated thin scintillator rods, according to EBAMed. Combining the positional information from two independent scintillator layers provides more accurate signal readouts compared to existing solutions.
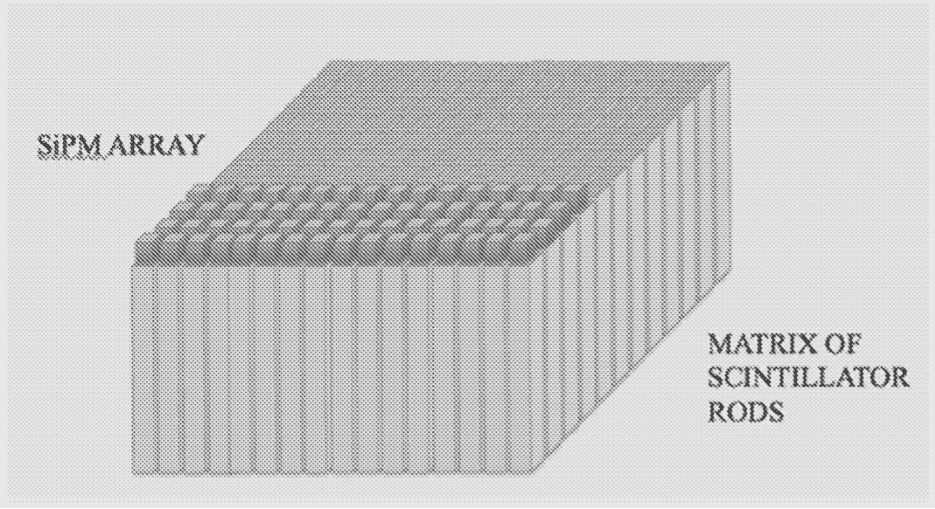
TAn example of a scintillator rod matrix with a silicon photomultiplier (SiPM) matrix reading mechanism.
The first module receives the gamma rays coming from a patient during hadron therapy and generates an electromagnetic shower. The first module is thin enough to transmit the electromagnetic shower to the second module, which is thick enough to absorb practically all of the electromagnetic energy.
At least one of the modules would direct the scintillation light to suitable optical sensors. The second module would provide a quantitative measurement of the energy released by the incoming radiation.
EBAMed says Wavelength Shifter strips in contact with opposite faces of the scintillator stacks provide the analog recording of signals, which are then converted into two-dimensional projective readout by an array of silicon photomultipliers.
The featured patent application, “Prompt Gamma Monitor for Hadron Therapy”, was filed with the USPTO on October 10, 2019 and published thereafter on September 16, 2021. The listed applicant is EBAMed SA. The listed inventors are Fabio Sauli and Adriano Garonna.